weic2303 — Science Release
Webb Unveils Dark Side of Pre-stellar Ice Chemistry
23 January 2023
The discovery of diverse ices in the darkest, coldest regions of a molecular cloud measured to date has been announced by an international team of astronomers using the NASA/ESA/CSA James Webb Space Telescope. This result allows astronomers to examine the simple icy molecules that will be incorporated into future exoplanets, while opening a new window on the origin of more complex molecules that are the first step in the creation of the building blocks of life.
If you want to build a habitable planet, ices are a vital ingredient as they are the main carriers of several key light elements — namely carbon, hydrogen, oxygen, nitrogen, and sulphur (referred to collectively as CHONS). These elements are important ingredients in both planetary atmospheres and molecules like sugars, alcohols, and simple amino acids. In our Solar System, it is thought they were delivered to Earth’s surface by impacts with icy comets or asteroids. Furthermore, astronomers believe such ices were most likely already present in the dark cloud of cold dust and gas that would eventually collapse to make the Solar System. In these regions of space, icy dust grains provide a unique setting for atoms and molecules to meet, which can trigger chemical reactions that form very common substances like water. Detailed laboratory studies have further shown that some simple prebiotic molecules can form under these icy conditions.
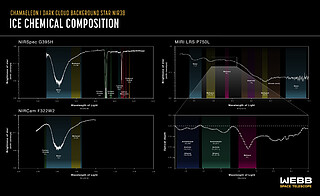
Now an in-depth inventory of the deepest, coldest ices measured to date in a molecular cloud [1] has been announced by an international team of astronomers using the NASA/ESA/CSA James Webb Space Telescope. In addition to simple ices like water, the team was able to identify frozen forms of a wide range of molecules, from carbonyl sulfide, ammonia, and methane, to the simplest complex organic molecule, methanol (in the interstellar medium, organic molecules are considered to be complex when having six or more atoms). This is the most comprehensive census to date of the icy ingredients available to make future generations of stars and planets, before they are heated during the formation of young stars. These icy grains grow in size as they are funnelled into the protoplanetary discs of gas and dust around these young stars, essentially allowing astronomers to study all the potential icy molecules that will be incorporated into future exoplanets.
“Our results provide insights into the initial, dark chemistry stage of the formation of ice on the interstellar dust grains that will grow into the centimetre-sized pebbles from which planets form in discs,” said Melissa McClure, an astronomer at Leiden Observatory who is the principal investigator of the observing program and lead author of the paper describing this result. “These observations open a new window on the formation pathways for the simple and complex molecules that are needed to make the building blocks of life.”
In addition to the identified molecules, the team found evidence for prebiotic molecules more complex than methanol in these dense cloud ices, and, although they didn't definitively attribute these signals to specific molecules, this proves for the first time that complex molecules form in the icy depths of molecular clouds before stars are born.
“Our identification of complex organic molecules, like methanol and potentially ethanol, also suggests that the many star and planet systems developing in this particular cloud will inherit molecules in a fairly advanced chemical state,” added Will Rocha, an astronomer at Leiden Observatory who contributed to this discovery. “This could mean that the presence of prebiotic molecules in planetary systems is a common result of star formation, rather than a unique feature of our own Solar System.”
By detecting the sulfur-bearing ice carbonyl sulfide, the researchers were able to estimate the amount of sulfur embedded in icy pre-stellar dust grains for the first time. While the amount measured is larger than previously observed, it is still less than the total amount expected to be present in this cloud, based on its density. This is true for the other CHONS elements as well. A key challenge for astronomers is understanding where these elements are hiding: in ices, soot-like materials, or rocks. The amount of CHONS in each type of material determines how much of these elements end up in exoplanet atmospheres and how much in their interiors.
“The fact that we haven't seen all of the CHONS that we expect may indicate that they are locked up in more rocky or sooty materials that we cannot measure,” explained McClure. “This could allow a greater diversity in the bulk composition of terrestrial planets.”
The ices were detected and measured by studying how starlight from beyond the molecular cloud was absorbed by icy molecules at specific infrared wavelengths visible to Webb. This process leaves behind chemical fingerprints known as absorption spectra which can be compared with laboratory data to identify which ices are present in the molecular cloud. In this study, the team targeted ices buried in a particularly cold, dense and difficult to investigate region of the Chameleon I molecular cloud, a region 631 light-years from Earth which is currently in the process of forming dozens of young stars.
“We simply couldn't have observed these ices without Webb,” elaborated Klaus Pontoppidan, Webb project scientist at the Space Telescope Science Institute, who was involved in this research. “The ices show up as dips against a continuum of background starlight. In regions that are this cold and dense, much of the light from the background star is blocked and Webb’s exquisite sensitivity was necessary to detect the starlight and therefore identify the ices in the molecular cloud.”
This research forms part of the Ice Age project, one of Webb's 13 Early Release Science programs. These observations are designed to showcase Webb’s observing capabilities and allow the astronomical community to learn how to get the best from its instruments. The Ice Age team have already planned further observations, and hope to trace out the journey of ices from their formation through to the assemblage of icy comets.
“This is just the first in a series of spectral snapshots that we will obtain to see how the ices evolve from their initial synthesis to the comet-forming regions of protoplanetary discs,” concluded McClure. “This will tell us which mixture of ices — and therefore which elements — can eventually be delivered to the surfaces of terrestrial exoplanets or incorporated into the atmospheres of giant gas or ice planets.”
Notes
[1] A molecular cloud is a vast interstellar cloud of gas and dust in which molecules can form, such as hydrogen and carbon monoxide. Cold, dense clumps in molecular clouds with higher densities than their surroundings can be the sites of star formation if these clumps collapse to form protostars.
More information
Webb is the largest, most powerful telescope ever launched into space. Under an international collaboration agreement, ESA provided the telescope’s launch service, using the Ariane 5 launch vehicle. Working with partners, ESA was responsible for the development and qualification of Ariane 5 adaptations for the Webb mission and for the procurement of the launch service by Arianespace. ESA also provided the workhorse spectrograph NIRSpec and 50% of the mid-infrared instrument MIRI, which was designed and built by a consortium of nationally funded European Institutes (The MIRI European Consortium) in partnership with JPL and the University of Arizona.
Webb is an international partnership between NASA, ESA and the Canadian Space Agency (CSA).
The international team of astronomers in this study consists of M. K. McClure (Leiden Observatory, Leiden University), W. R. M. Rocha (Laboratory for Astrophysics, Leiden Observatory), K. M. Pontoppidan (Space Telescope Science Institute), N. Crouzet (Leiden Observatory, Leiden University), L. E. U. Chu (NASA Postdoctoral Program Fellow, NASA Ames Research Center), E. Dartois (Institut des Sciences Moleculaires d’Orsay (ISMO), CNRS/Univ. Paris-Saclay), T. Lamberts (Leiden Institute of Chemistry, Gorlaeus Laboratories & Leiden Observatory, Leiden University), J. A. Noble (Physique des Interactions Ioniques et Moléculaires (PIIM), CNRS/Aix-Marseille Univ.), Y. J. Pendleton (Department of Physics, University of Central Florida), G. Perotti (Max Planck Institute for Astronomy), D. Qasim (Southwest Research Institute, San Antonio, TX, USA.), M. G. Rachid (Laboratory for Astrophysics, Leiden Observatory), Z. L. Smith (School of Physical Sciences, The Open University), F. Sun (Steward Observatory, University of Arizona), Tracy L. Beck (Space Telescope Science Institute), A. C. A. Boogert (Institute for Astronomy, University of Hawai‘i at Manoa), W. A. Brown (Department of Chemistry, University of Sussex), P. Caselli (Max Planck Institute for Extraterrestrial Physics), S. B. Charnley (Solar System Exploration Division, NASA Goddard Space Flight Center), Herma M. Cuppen (Institute for Molecules and Materials, Radboud University), H. Dickinson (School of Physical Sciences,The Open University), M. N. Drozdovskaya (Center for Space and Habitability, University of Bern), E. Egami (Steward Observatory, University of Arizona), J. Erkal (Institute for Astronomy, University of Hawai‘i at Manoa), H. Fraser (School of Physical Sciences, The Open University), R. T. Garrod (Departments of Astronomy & Chemistry, University of Virginia), D. Harsono (Institute of Astronomy, Department of Physics, National Tsing Hua University), S. Ioppolo (Center for Interstellar Catalysis, Department of Physics and Astronomy, Aarhus University), I. Jiménez-Serra (Centro de Astrobiología [CAB], Ctra. de Ajalvir), M. Jin (Solar System Exploration Division, NASA Goddard Space Flight Center & Department of Physics, Catholic University of America), J. K. Jørgensen (Niels Bohr Institute, University of Copenhagen), L. E. Kristensen (Niels Bohr Institute, University of Copenhagen), D. C. Lis (Jet Propulsion Laboratory, California Institute of Technology), M. R. S. McCoustra (Institute of Chemical Sciences, Heriot-Watt University), Brett A. McGuire (Department of Chemistry, Massachusetts Institute of Technology and National Radio Astronomy Observatory), G. J. Melnick (Center for Astrophysics | Harvard & Smithsonian), Karin I. Öberg (Center for Astrophysics | Harvard & Smithsonian), M. E. Palumbo (INAF - Osservatorio Astrofisico di Catania), T. Shimonishi (Faculty of Science, Niigata University), J. A. Sturm (Leiden Observatory, Leiden University), E. F. van Dishoeck (Leiden Observatory, Leiden University), and H. Linnartz (Laboratory for Astrophysics, Leiden Observatory).
Image Credit: NASA, ESA, CSA, and M. Zamani (ESA/Webb); Science: F. Sun (Steward Observatory), Z. Smith (Open University), and the Ice Age ERS Team.
Links
- ESA Webb Seeing Farther Interactive Brochure
- Early Release Science Program 1309: IceAge: Chemical Evolution of Ices during Star Formation
- Release on STScI website
- Release on NASA website
- Release on ESA website
- Science paper
Contacts
Melissa McClure
Leiden University, The Netherlands
Email: [email protected]
Bethany Downer
ESA/Webb Chief Science Communications Officer
Email: [email protected]
Ninja Menning
ESA Newsroom and Media Relations Office
Email: [email protected]